Discover what healthcare professionals (HCPs) think about pharmaceutical products and their manufacturers, as it happens, through CREATION.co’s tracking updates. How are HCPs responding to the latest trial results for COVID-19 vaccine candidates? How are HCPs talking about or engaging with the Top 50 pharma companies on social media? Each week CREATION.co’s tracking updates bring you the latest insights from the conversation of HCPs across the globe discussing these topics and more.

The Top 10 covid-19 vaccine candidates mentioned by HCPs on Twitter in August
The top 10 most mentioned COVID-19 vaccine candidate list by HCPs was supplemented with the new vaccines by Gamaleya Research Institute, Curevac anda Sanofi and GSK collaboration, while the top 4 candidates upheld their position despite a change in the most mentioned COVID-19 vaccine candidate.
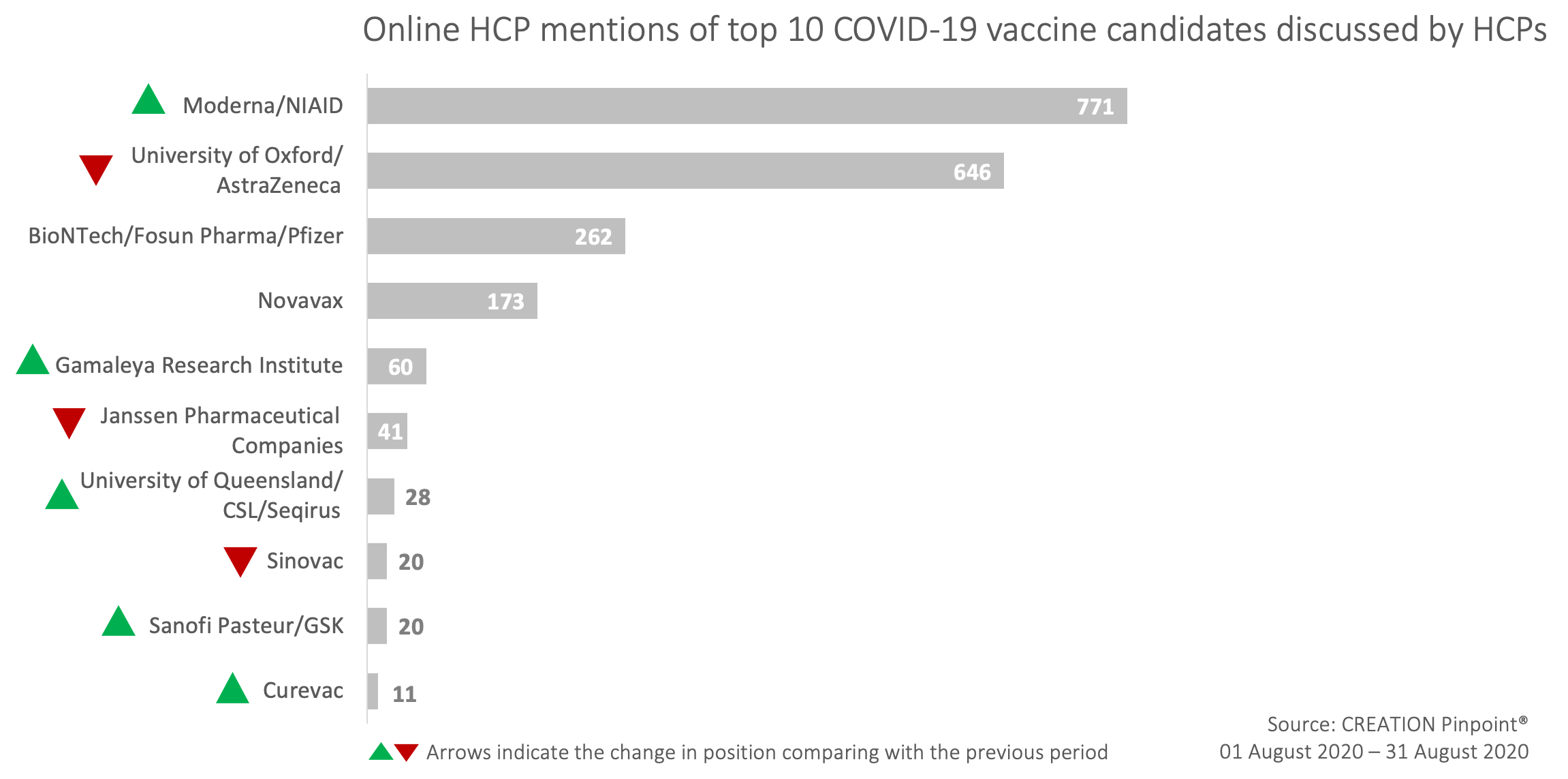
August’s insights from HCPs mentioning covid-19 vaccine candidates on Twitter
During the August 2020 HCP conversation around COVID-19 vaccine candidates in clinical evaluation has decreased, however HCPs have produced three times more original content compared to July 2020.
If during July 2020 the online HCP discussion on Twitter was driven by the study data and new trial stage announcements, August has shifted HCP focus from the scientific advancements to the practicalities of delivering vaccines to people.

The month started with HCPs picking up on Moderna’s announcement to charge between 32$ and 37$ for a potential vaccine. HCPs shared posts and articles highlighting that the vaccine was already funded by taxpayer money and compared it to the $19.95 price of Pfizer candidate. A physician in the US produced a Twitter thread sharing various media sources on the topic and called it “paying twice for a vaccine”.
#BigPharma uses research funded by #Taxpayers through the @NIH and then charges the American people the highest possible drug prices. #NIH–#Moderna investigational #COVIDー19 vaccine shows promise in mouse studies https://t.co/GFSbEPsftz #CoronavirusPandemic #PandemicProfiteers
— Dana Fairbanks, MD #AaronBushnell #FreePalestine (@dcfairbank) August 5, 2020
Another large portion of the online HCP dialogue around the COVID-19 vaccine candidates focused on two countries in the process of agreements for supplying the vaccine – Australia and Canada.
A misunderstanding about a deal between AstraZeneca and the Australian government caught HCP attention, who at first shared the announcement of the “secured deal”, only then to come across conflicting statements by AstraZeneca’s about incomplete agreement awaiting further steps.
https://twitter.com/KKeneally/status/1295957671355363328
A recent agreement in Canada for the supply of Novavax vaccine has stirred a more positive HCP discussion. HCPs on Twitter, like Infectious Disease physician Isaac Bogoch, shared the good news of the country now having secured access to several different key vaccine candidates with three different mechanisms of action.
Canada now has a deal with Novavax for 76 million doses of a #COVID19 vaccine.
Now we have vaccine agreements with Moderna, Pfizer, and Novavax.
Great news.
Fingers now tightly crossed for successful Phase 3 clinical trials.https://t.co/omCGHJdbhH
— Isaac Bogoch (@BogochIsaac) August 31, 2020
We will be tracking the online HCP conversation to identify trends and change in views relating to COVID-19 vaccines. You can stay up to date with HCP insights by subscribing to CREATION.co’s monthly eJournal.
- Data for this research was analysed using CREATION Pinpoint® from the online Twitter conversations of HCPs around the world in English language (other languages are available), between August 1st – August 31st, 2020.
- Vaccines tracked were the 36 COVID-19 vaccine candidates in clinical evaluation before 21st September 2020 (WHO Draft landscape of COVID-19 candidate vaccines).

 By Laura McIntyre
By Laura McIntyre 
