Cancerous tumours in the brain and central nervous system (CNS) is the 10th leading cause of death for men and women worldwide, with an estimated 251,329 people dying from primary cancerous brain and CNS tumours in 2020 according to ASCO’s patient information website. Despite the severity of the condition, there is still only a limited understanding of the direct causes of brain tumours and only a limited range of treatment options available for HCPs to use.
While HCP conversation around brain tumours was active consistently throughout the last 12 months, the volume of discussion surrounding it increased during relevant awareness days and months, as well as during the annual conference of the European Association of Neuro-Oncology.
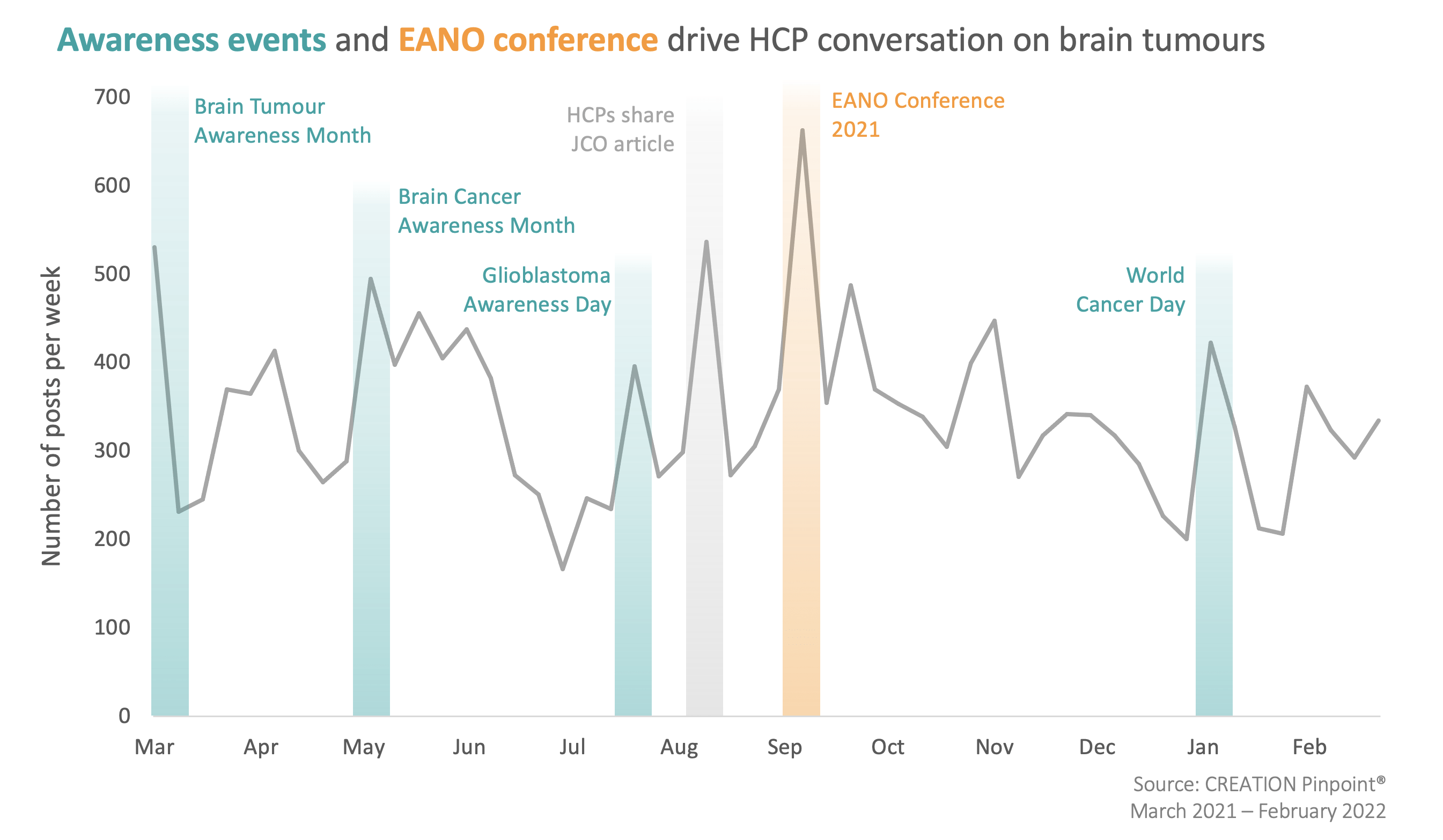
As CREATION.co has previously identified, HCPs regularly engage with disease awareness days in order to draw attention to their specific therapy area, often highlighting the frustrations and difficulties that they and their patients experience. In the brain tumour space, the key concerns that HCPs frequently expressed centred around a lack of pharmaceutical products approved to treat brain cancer, as well as the lack of progress in effective treatment of the disease.
Treatment options for glioblastoma, and all brain cancers, are extremely limited. There are only 5 drugs and 1 device approved by the FDA to treat brain tumors. Learn what treatment was like for one GBM patient, @DJStewart816. #GBMDay #braintumor #btsm https://t.co/8Km8nBf7oW
— Head For The Cure (@HeadfortheCure) July 21, 2021
May is Brain Tumor Awareness Month.
Sure, we’re aware of cancer. But the FDA has only approved five medications and one device to treat brain cancer—and life expectancy has only improved a dismal three months in the last three decades.
Doctors don’t even know what causes it.
— Anastasia Golovashkina (@golovashkina) May 1, 2021
The Brain Tumour Social Media (#BTSM) chat, which hosts monthly discussions on Twitter on brain tumour related topics, further highlighted this issue – noting how little development there had been in terms of new therapies of the past 40 year period, as well as sharing an image which highlighted HCPs dissatisfaction with the current options available to them.
Truth.
Over the last 40 years, just 5 therapies have been approved by the @US_FDA to treat primary brain tumors. #btsm https://t.co/Gzvt6YeGtu
— #BTSM chat (@BTSMchat) March 28, 2021
HCPs want more treatments options
Of the treatment options currently available, temozolomide was the most frequently mentioned therapy by HCPs, and has been shown in clinical trials to improve overall survival for those with malignant brain tumours. HCPs acknowledged that this treatment is currently the best option available, though in some cases due to the presence of a DNA repair gene known as MGMT it can still be ineffective. However, a lack of alternatives often means that HCPs will proceed with temozolomide regardless of the presence of MGMT.
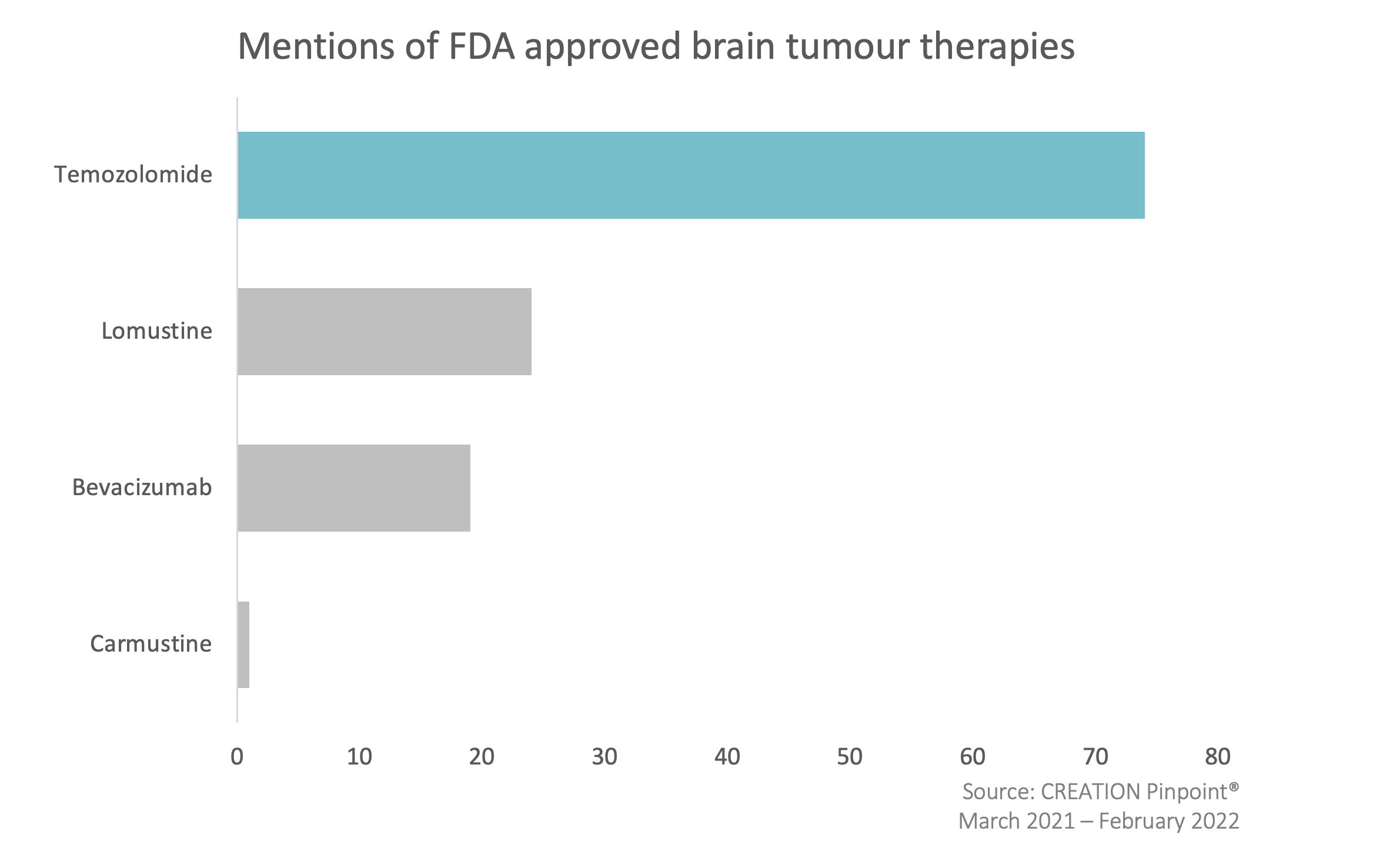
When mentioning other treatments, Lomustine was frequently highlighted by HCPs for its increasing cost, with the current price being roughly 1,900% than it was 8 years ago; while bevacizumab was seen to have had disappointing trial results. Together, this demonstrates HCPs frustration with all of the current options available to them and illustrates the need for new treatment options in order to improve the outcomes for brain tumour patients.
💸 @OncoAlert: "Since taking over manufacturing rights, NextSource has become the single-source manufacturer of lomustine in the US… Today, NextSource’s list price is ~1,900% higher than it was just 8 years ago" #pricegauging #btsm #financialToxicity https://t.co/sazWZxgh7k pic.twitter.com/THlwouKqhY
— Fumiko Ladd Chino, MD (@fumikochino) July 26, 2021
@BrainTumorDoc gives an update on his BEV + immuno work -> Randomized phase 2 study of nivolumab (nivo) + either standard or reduced dose bevacizumab (bev) in recurrent glioblastoma (rGBM). Another disappointing therapy #Btsm #asco21 pic.twitter.com/IvKhyWHFQU
— Ashley Love Sumrall, MD, FACP, FASCO (@AshleySumrallMD) June 6, 2021
HCPs campaign for more research funding
One of the key factors limiting progress in treatment of brain cancers has been the lack of funding for researchers working in the space. Brain tumours are particularly difficult to treat effectively due to the blood-brain barrier, which many typical cancer therapies cannot cross in order to target the cancer. As such, HCPs across various nations have taken to social media in order to try and address this problem, campaigning for additional funding into brain tumour research and treatment. In the US, HCPs joined the National Brain Tumor Society in urging their local political representatives to increase research funding and healthcare access.
In Australia, a neurosurgeon drew attention to the current 100% mortality rate of glioblastoma, the most common type of brain tumour in adults, as evidence that more research into new treatments is needed.
https://twitter.com/sammons_vanessa/status/1477818927979794433
And in the UK, HCPs shared charitable organisations that use donations to fund brain tumour research, as well as petitioning leaders of their government.
#RareDiseaseDay2022 I work with various rare tumour groups #acousticneuroma #chordoma #chondrosarcoma #nf2 to name a few. More funds are needed to advance research/treatment. Follow/donate to these great charities @BANAUK @ChordomaUK @Sarcoma_UK @BrainTumourOrg @nf2biosolution1
— Andrea wadeson (@mamawadey) February 28, 2022
HCPs continue working with limited resources
Over the last year, the difficulties already facing HCPs in their efforts to treat brain tumours have been further heightened by the additional burden of COVID-19, meaning that for many patients their treatments had to be delayed. Many HCPs lamented this situation, sharing stories of patients whose surgery had been delayed.
Do you remember the man in December (I think) sharing the scan of his brain tumor??? He had his brain tumor surgery cancelled because of the burden of Covid on his hospital. I think about him a lot. I hope he is ok.
— Jennifer Gunter (@DrJenGunter) February 4, 2022
Nonetheless, HCPs and researchers have continued to persevere in the face of difficult circumstances, using the funding available to them to continue to search for better therapy options to treat brain tumours and ways to increase the survival chances of patients.
This is what real #superheroes look like. #labmeeting to find better #GBM #braintumor therapy. Incredibly honored to work with the brightest @TXMedCenter @MDAndersonNews @BettyKimLab @wenjiang_nano #endcancer #oncsurgery #neurosurgery pic.twitter.com/zKiSskl1pY
— Betty Y.S. Kim, MD, PhD, FRCSC, FAIMBE (@BettyKimLab) February 10, 2022
The EANO congress offered some great examples of the advancements HCPs and researchers have been making in brain tumour treatment. Some of the highlights included a new DNA damage response biology generating new treatments, in-depth explorations of the blood brain barrier, and presentations on the clinical implications of genetic scars induced by radiotherapy.
Award winning data on genetic scar induced by radiotherapy in #glioblastoma and its potential clinical implications presented by @ekocakavuk at #EANO2021. Session chair @valientelab notes that findings may also be relevant in #brainmetastases, research ongoing. #btsm @OncoAlert pic.twitter.com/BJs5bnS8pK
— EANO (@EANOassociation) September 26, 2021
Throughout the last year, HCPs have used social media as a tool to amplify their concerns around brain tumour research and treatment, calling for increased investment into exploring new and improved therapy options to be able to offer patients. Additionally, social media offered an opportunity for HCPs to share the progress that was being made, and celebrate those contributing to brain tumour research.
If you are interested in finding out more about the needs of HCP communities within a specific therapy area, you can register for CREATION’s monthly knowledge e-journal, or get in touch with us directly to see how we can support your work.

 By Paul Cranston
By Paul Cranston