At CREATION we have been tracking how US healthcare professionals (HCPs) discussed pancreatic cancer online during the American Society of Clinical Oncology’s (ASCO) annual meeting. Within this conversation, a big talking point among HCPs was the release of the POLO trial results.
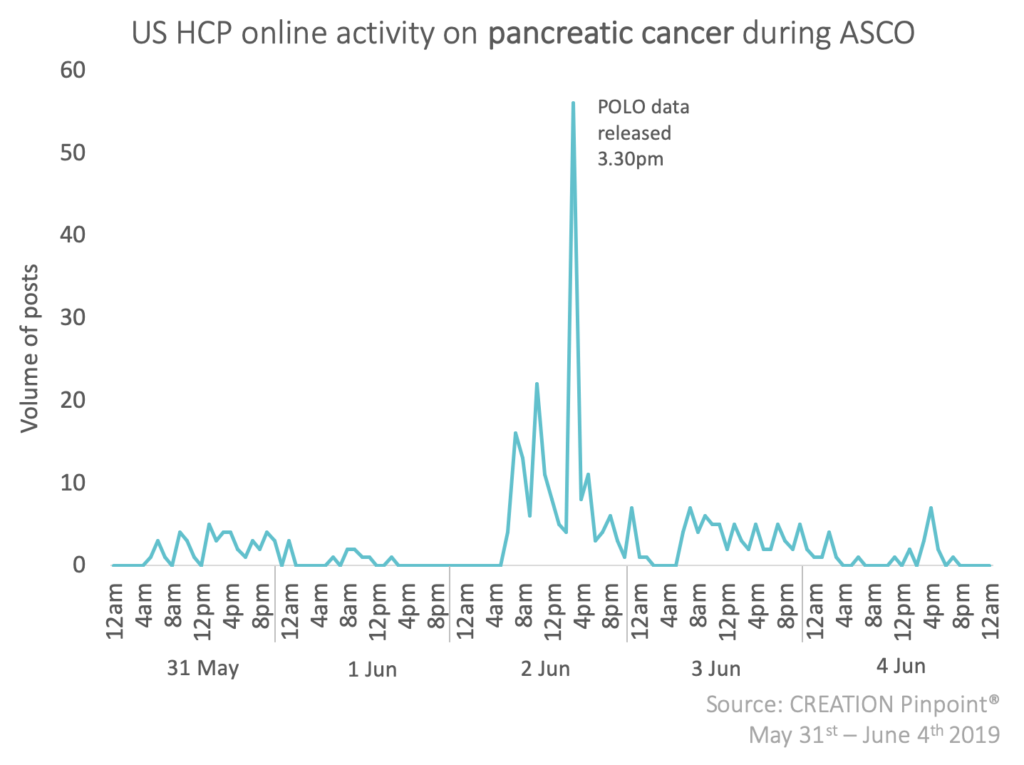
The POLO trial is a phase 3 trial looking at AstraZeneca and Merck’s (MSD) product, olaparib, for pancreatic cancer that has spread in people with a BRCA genetic mutation.
When discussing the POLO trial, US HCPs used words like ‘practice-changing’ to describe their positivity towards the progression-free survival results, however, some HCPs shared concerns about the cost and overall survival rate compared to placebo.
Following the release of the POLO trial data, US HCPs used social media to encourage genetic testing to identify patients who have a risk of BRCA pancreatic cancer mutations. A post by David Solit, an oncologist, highlighted that HCPs need to implement genetic testing for all pancreatic cancer patients.
US HCPs shared posts from and mentioned patient advocacy groups (PAGs). The Pancreatic Cancer Action Network and Let’s Win! Pancreatic Cancer were the two most shared PAGs at ASCO19 in the pancreatic cancer conversation. A video series from Let’s Win about patient treatment journeys and educational content about treatment in collaboration with PeerView were shared by a number of HCPs.
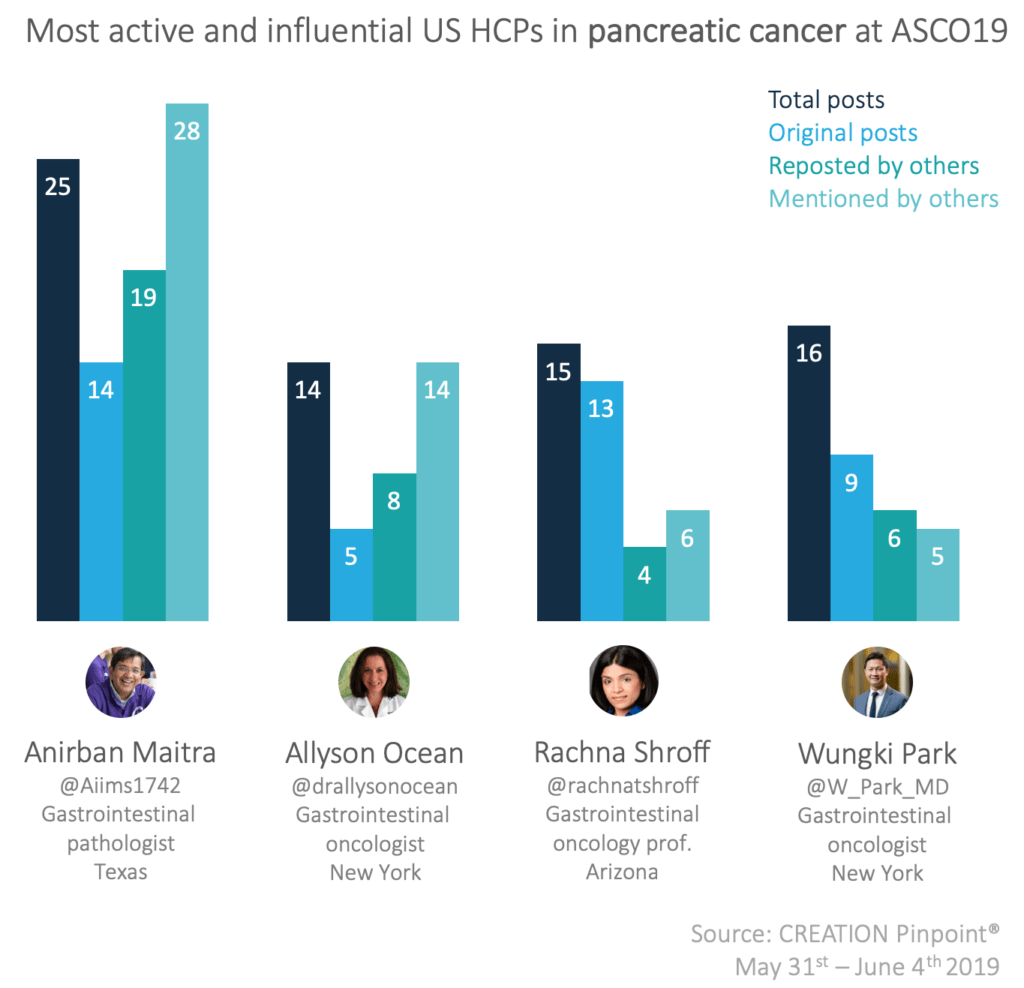
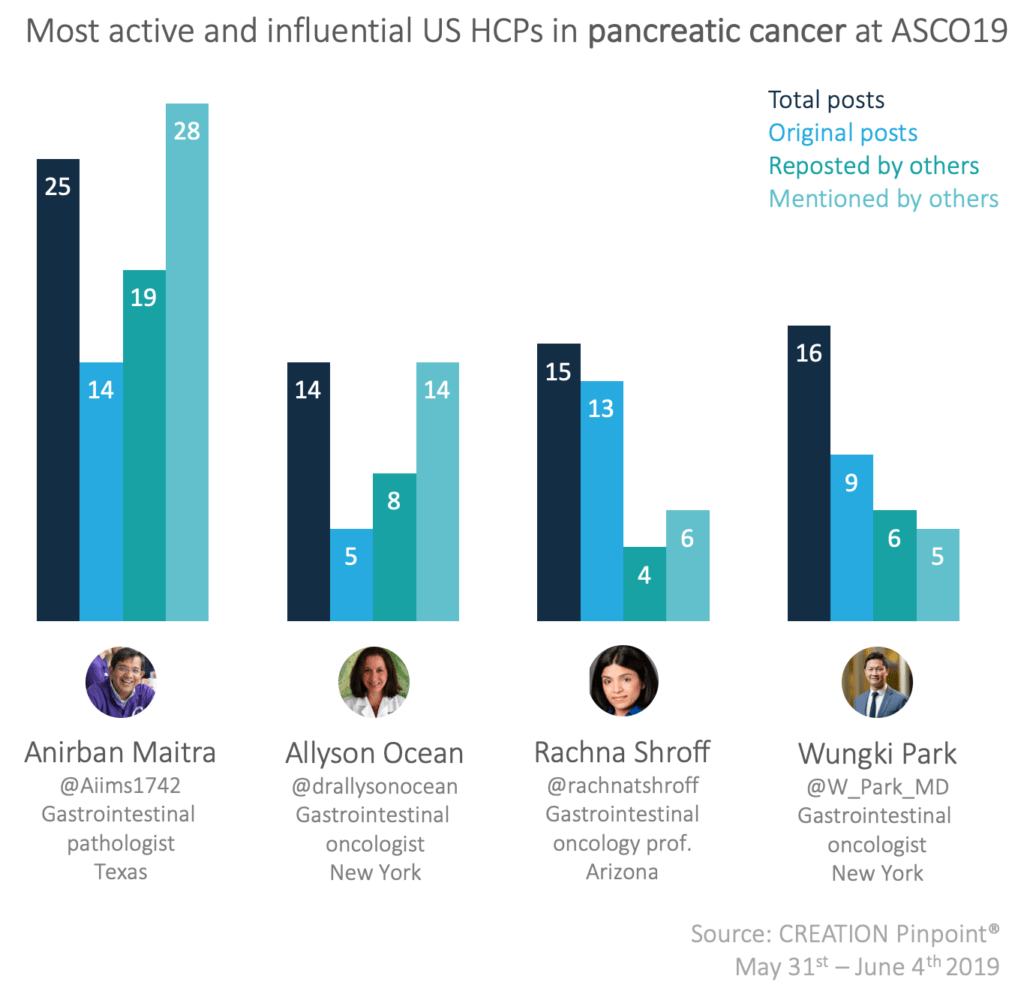
The top US HCPs in the pancreatic cancer conversation at ASCO19 were both active online and were well engaged with by their peers online.
A study of HCP views on pancreatic cancer diagnosis and treatment in the US was recently completed using CREATION Pinpoint®. These insights follow on from that study in which we will continue to bring tracking updates. To access the study, other resources and receive tracking updates of the pancreatic cancer conversation by US HCPs, sign up here.